The landscape of medical device tender strategy in Malaysia has shifted. On January 28, 2026, the Medical Device Authority (MDA) released Circular Letter No. 1/2026, a policy that ensures every supplier is following the same best-practice rules to keep patients safe.
Taking action now is your best path to securing new opportunities in government and private healthcare. Here is everything you need to know about the end of exemptions and the new mandatory path to market access.
No more medical device tender without Establishment License as Importer or Distributor
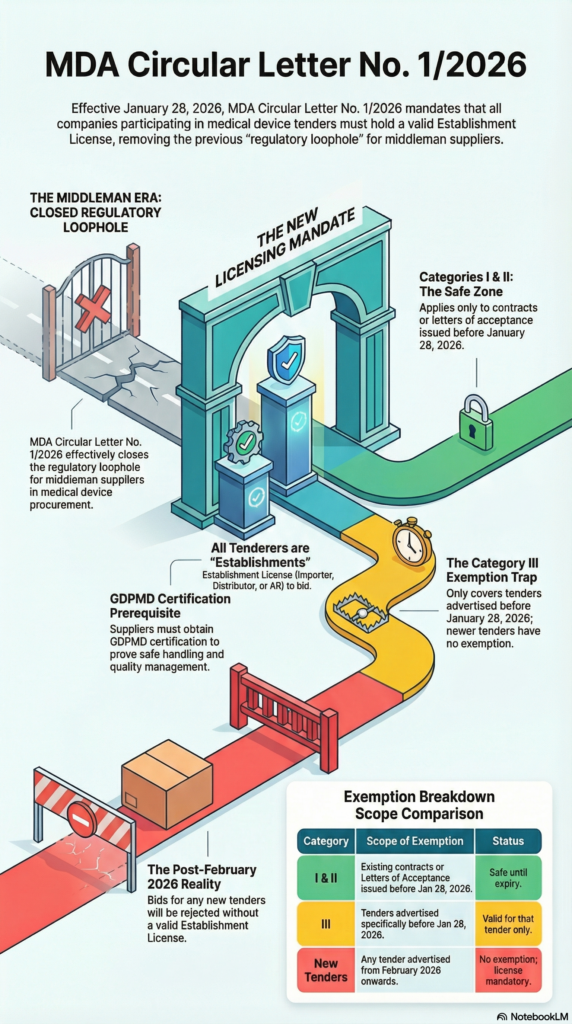
Historically, medical device tender strategy are in such the tenderers acted merely as middleman suppliers without holding their own establishment licenses. However, under Circular 1/2026, this is no longer applicable.
The MDA now officially classifies all tenderers as “Establishments.” Whether you are an Importer, a Distributor, or an Authorized Representative (AR), you are now legally required to hold a valid Establishment License in your company’s name to even submit a bid.
Understanding the Category III Exemption
Hence, to prevent a total supply chain freeze, the MDA introduced three categories of exemptions. Anyhow, many businesses are now making the smart switch from temporary exemptions to permanent, long-term success.
- Category I & II: Cover contracts already awarded or Letters of Acceptance (LOA) issued before January 28, 2026. Thus, these are safe until the contract expires.
- Category III: This applies to tenders that were advertised before January 28, 2026.
Unlock a world of new contracts! As we move past the temporary Exemption III stage, having your GDPMD and license in hand is the smartest way to show partners you are ready for business. Let’s get your credentials ready so you never miss a beat!
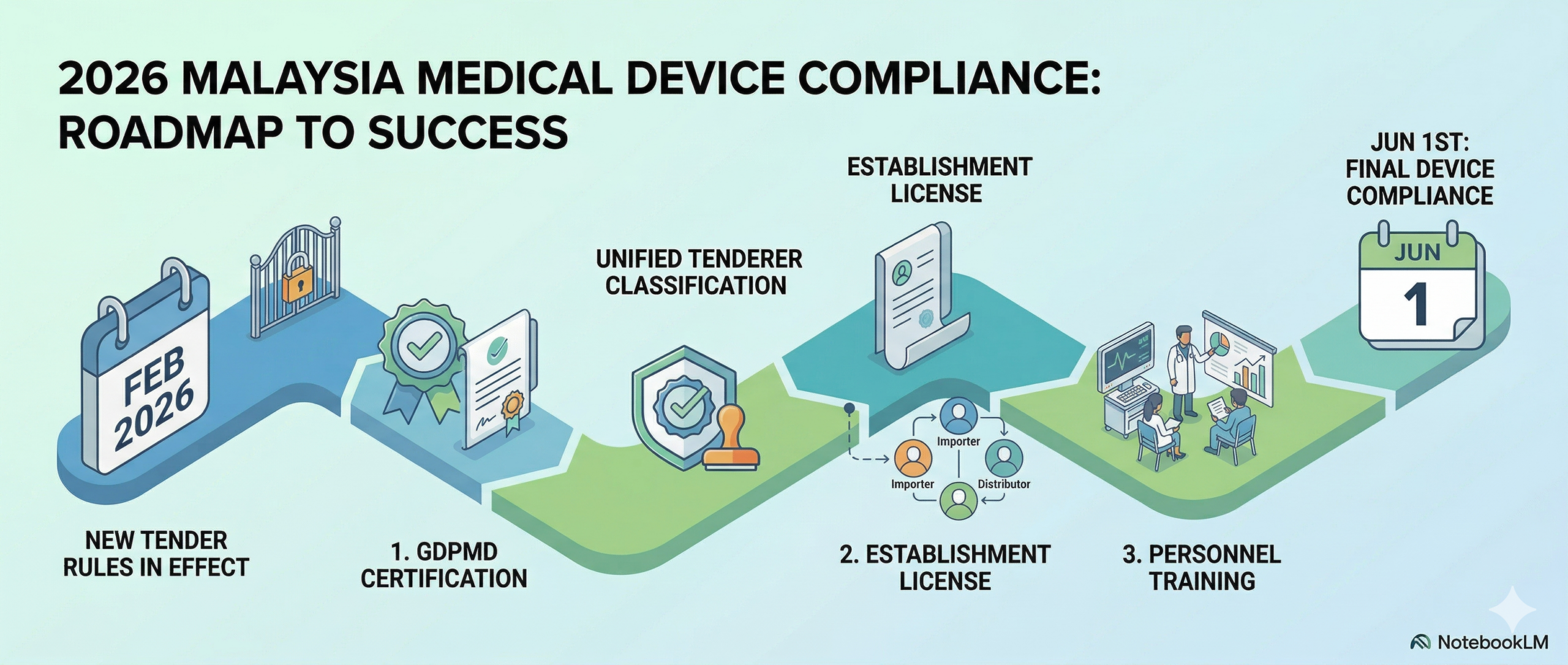
The 3-Step Medical Device Compliance Roadmap for 2026
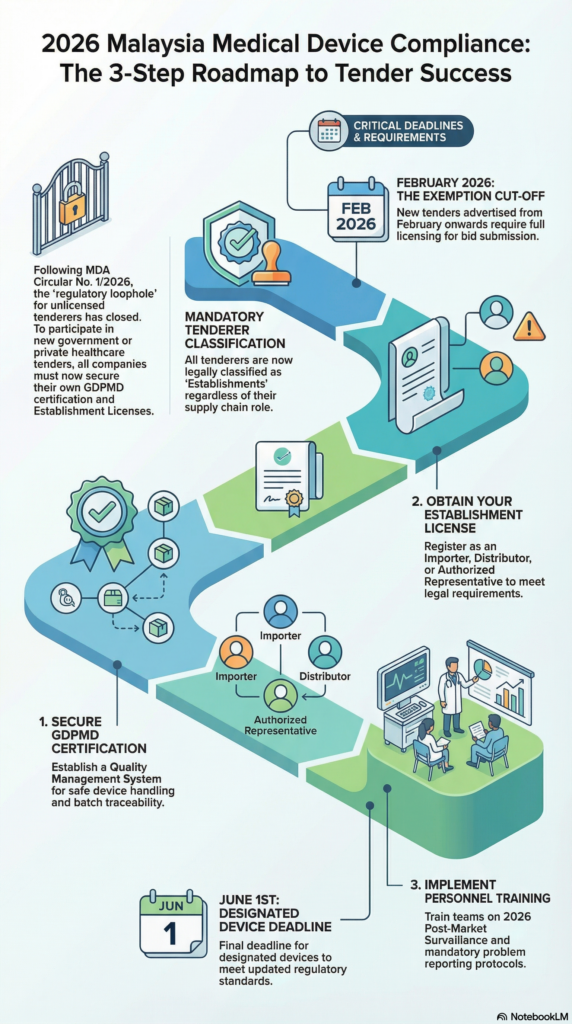
In a nutshell, to stay in the game, medical device tender strategy must shift through three critical phases:
1. Secure Your GDPMD Certification Good Distribution Practice for Medical Devices (GDPMD) is the prerequisite for your license. It proves your company has a Quality Management System (QMS) capable of handling medical devices safely—covering everything from storage temperatures to batch traceability.
2. Choose the Right Establishment License Are you an Importer bringing goods from overseas, a Distributor selling locally or an Authorized Representative (AR) acting on behalf of a foreign brand? Your license must accurately reflect your business model to satisfy Sections 9(a) and 9(c) of the new Circular.
3. Personnel Training Compliance isn’t just a certificate on the wall, but it’s a daily practice. With the MDA’s increased focus on Post-Market Surveillance (PMS) in 2026, your team must be trained on how to handle mandatory problem reporting and e-IFU (Electronic Instructions for Use) requirements.
How We Can Help
At Insyst TAC Sdn Bhd, we specialize in smooth compliance.
- GDPMD Consultation: We build your QMS from scratch and guide you through the audit.
- Establishment Licensing: We handle the paperwork for Importers, Distributors, and ARs.
- HRDCorp Claimable Training: Upskill your team on the latest 2026 MDA regulations without affecting your bottom line.
The June 1st deadline for designated devices is approaching, and new tenders are being advertised every week. Don’t let a missing license be the reason you lose your next contract. Contact us now to get more information on Establishment License!
